How is this drug name pronounced?
Ramucirumab:RA-myoo-SIR-yoo-mab
Cyramza: sy-RAM-zuh
What cancer(s) does this drug treat?
Cyramza has been approved for a number of cancer types and stages, in some cases as a single therapy and in some cases in combination with other therapies.
Cyramza is approved for:
Stomach cancer
Non-small cell lung cancer
Colorectal cancer
Liver cancer
Advanced stomach cancer
Cyramza is approved for:
- Patients with advanced gastric adenocarcinoma (stomach cancer) or gastro-esophageal junction adenocarcinoma (cancer of the area where the stomach meets the esophagus) that has spread to other parts of the body, and who have been treated with chemotherapy containing platinum or fluoropyrimidine, and it either did not work or stopped working. In such cases, Cyramza may be used by itself or in combination with paclitaxel (Taxol or Onxal).
Advanced non-small cell lung cancer
Cyramza is approved for:
- Patients with advanced non-small cell lung cancer that has spread to other parts of the body, and who have been treated with chemotherapy containing platinum, and it either did not work or stopped working. In such cases, Cyramza is used in combination with docetaxel (Docefrez or Taxotere). If the patient’s tumor has an abnormal EGFR or ALK gene, the patient has to have also tried an approved therapy for tumors with such abnormal genes prior to receiving treatment containing Cyramza.
- Patients with advanced non-small cell lung cancer that has spread to other parts of the body, and whose tumors test positive for certain activating mutations of the EGFR protein (exon 19 deletions or exon 21 (L858R) substitutions). In such cases, Cyramza is used in combination with erlotinib (Tarceva) as a first treatment.
Advanced colorectal cancer
Cyramza is approved for:
- Patients with advanced colon or rectal cancer that has spread to other parts of the body, and who have received therapy containing bevacizumab (Avastin, Mvasi, Zirabev), oxaliplatin (Eloxatin), and fluoropyrimidine, and it either did not work or stopped working. In such cases, Cyramza is used in combination with irinotecan (Onivyde or Camptosar), folinic acid, and fluorouracil.
Advanced liver cancer
Cyramza is approved for:
- Patients with advanced hepatocellular carcinoma (liver cancer) who have high levels of alpha fetoprotein in their blood, and who have been treated with sorafenib (Nexavar).
Limitations of use:
Age: The safety and efficacy of Cyramza in patients under 18 years of age have not been established.
Fertility/Pregnancy/Breastfeeding: Cyramza can impair the ability to have children. Cyramza can cause harm to a fetus, and is not recommended for use during pregnancy. Pregnancy should be prevented for at least three months after the last dose of Cyramza. The risks associated with Cyramza during breastfeeding are not known and cannot be ruled out. Due to the potential for serious adverse reactions in the breastfed child, women are advised not to breastfeed during treatment and for at least two months after the last dose of Cyramza.
What type of immunotherapy is this?
How does this drug work?
- Target: vascular endothelial growth factor (VEGF) receptor
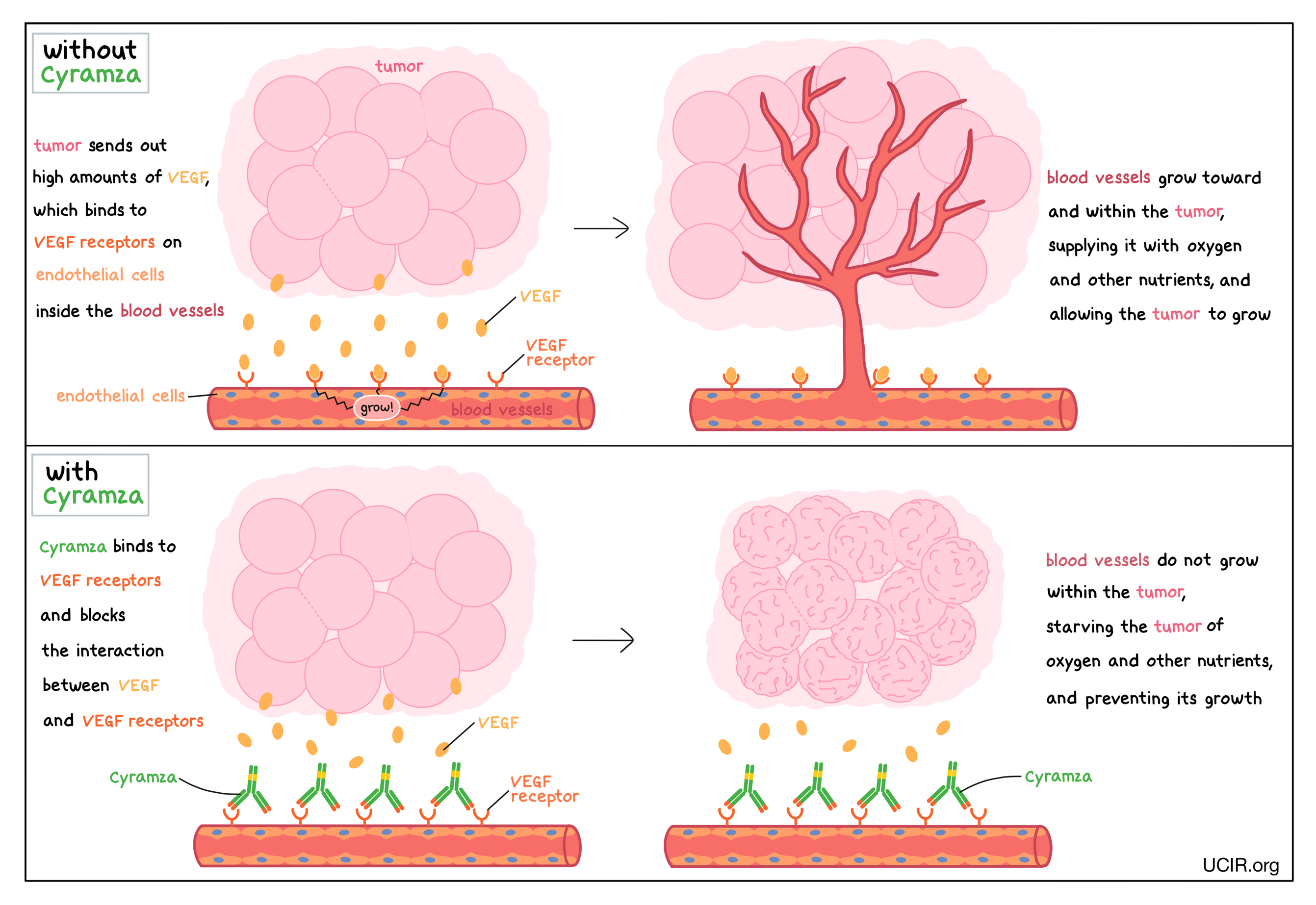
Cyramza is an antibody that attaches to protein molecules called vascular endothelial growth factor (VEGF) receptors, which are present on the surface of cells found in the inside lining of blood vessels (endothelial cells). VEGF molecules that freely float in tissues and in the blood bind to VEGF receptors, stimulating the formation of new blood vessels and promoting the growth of preexisting blood vessels – a process called angiogenesis. In a healthy person, VEGF molecules are produced by many different types of cells and help to create new blood vessels during embryonic development, normal growth of tissues, and wound healing. Tumors make higher than normal amounts of VEGF because the formation of new blood vessels within the tumor is critical for the tumor to grow (by providing the tumor with access to oxygen and other nutrients) and to spread to other parts of the body (by providing a route for cancer cells to enter blood circulation and move to other locations). In a patient with cancer, the binding of VEGF to VEGF receptors leads the endothelial cells to multiply, migrate to the source of the VEGF molecules (the tumor site), and form new blood vessels within the tumor.
Cyramza binds to the VEGF receptors in a way that blocks the interaction between VEGF receptors and VEGF. This helps prevent the formation of new blood vessels within the tumor, thus starving the tumor of oxygen and other nutrients, and preventing its growth.
How is this drug given to the patient?
Prior to receiving each dose of Cyramza, patients receive an antihistamine to reduce the chance of a reaction to the infusion. If an infusion-related reaction occurs, patients should receive an antihistamine, dexamethasone (or an equivalent corticosteroid), and acetaminophen prior to each subsequent dose of Cyramza.
Cyramza is administered via a tube into a vein (intravenous infusion, or I.V.) every two or three weeks (depending on the cancer type) and does not require a hospital stay. The first infusion is administered over 60 minutes. If the first infusion is tolerated, all following infusions are administered over 30 minutes.
What are the observed clinical results?
For:
Advanced stomach cancer
Advanced non-small cell lung cancer
Advanced colorectal cancer
Advanced liver cancer
It is important to keep in mind that each patient’s actual outcome is individual and may be different from the results found in the clinical studies. In addition, with immunotherapy, sometimes it takes several months for responses to be observed.
Advanced stomach cancer
In a clinical trial, 355 patients with advanced gastric cancer (stomach cancer) or gastro-esophageal junction adenocarcinoma (cancer of the area where the stomach meets the esophagus) that had grown or spread to other parts of the body, and who had been treated with chemotherapy containing platinum or fluoropyrimidine, and it either did not work or stopped working, were treated with either Cyramza or placebo. Patients treated with Cyramza lived for a median of 5 months, compared with 4 months for patients receiving placebo. For patients treated with Cyramza, the disease did not get worse for a median of 2 months, compared with 1 month for patients receiving placebo.
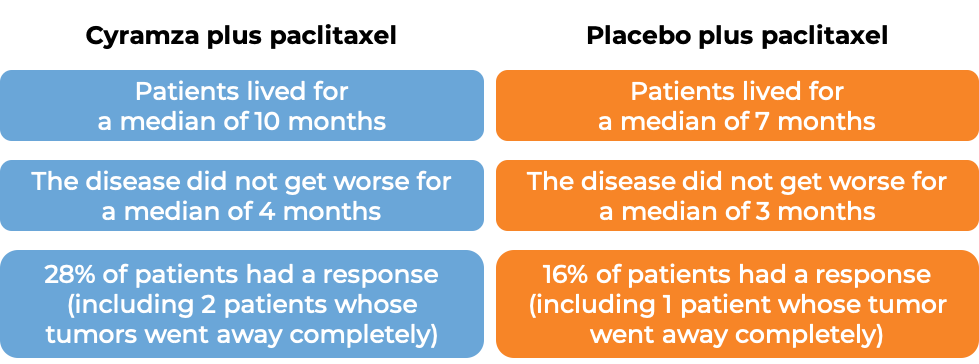
In another clinical trial, 665 patients with advanced gastric cancer or gastro-esophageal junction adenocarcinoma that had grown or spread to other parts of the body, and who had been treated with chemotherapy containing platinum or fluoropyrimidine, and it either did not work or stopped working, were treated with either:
- Cyramza plus paclitaxel (Taxol or Onxal), OR
- placebo plus paclitaxel.
After approximately two years, the following results were observed:
Advanced non-small cell lung cancer
In a clinical trial, 1253 patients with advanced non-small cell lung cancer that had grown or spread to other parts of the body, and who had been treated with chemotherapy containing platinum, and it either did not work or stopped working, were treated with either:
- Cyramza plus docetaxel (Docefrez or Taxotere), OR
- placebo plus docetaxel.
After approximately two and a half years, the following results were observed:

In another clinical trial, 449 patients with previously untreated advanced non-small cell lung cancer that had spread to other parts of the body, and whose tumors tested positive for certain activating mutations of the EGFR protein (exon 19 deletions or exon 21 (L858R) substitutions), were treated with either Cyramza plus erlotinib (Tarceva) OR placebo plus erlotinib (Tarceva). At a median follow-up of 21 months, the following results were observed:

Advanced colorectal cancer
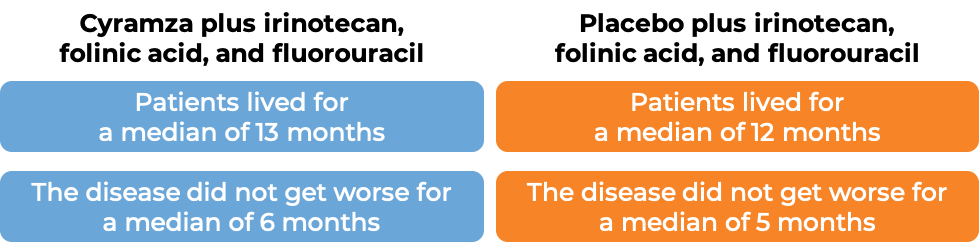
In a clinical trial, 1072 patients with colon or rectal cancer that had spread to other parts of the body, and who had received therapy containing bevacizumab (Avastin, Mvasi, Zirabev), oxaliplatin (Eloxatin), and fluoropyrimidine, and it either did not work or stopped working, were treated with either:
- Cyramza plus irinotecan (Onivyde or Camptosar), folinic acid, and fluorouracil, OR
- Placebo plus irinotecan, folinic acid, and fluorouracil.
Advanced liver cancer
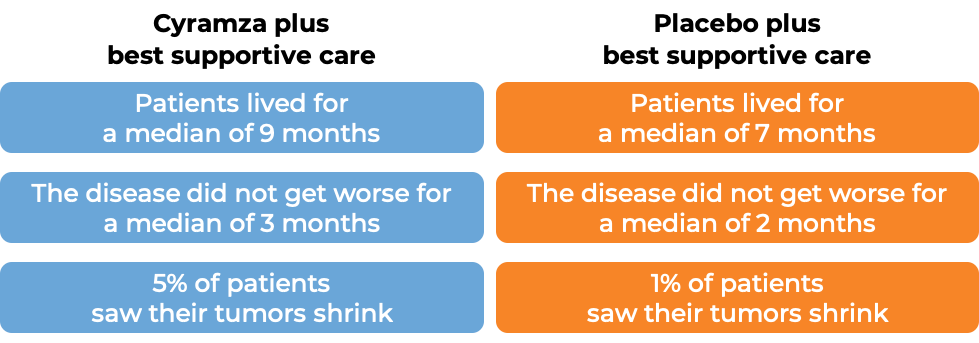
In a clinical trial, 292 patients with advanced hepatocellular carcinoma (liver cancer) who had high levels of alpha fetoprotein in their blood, and who had been treated with sorafenib (Nexavar), and it either did not work, stopped working, or could not be tolerated, were treated with either Cyramza plus best supportive care or placebo plus best supportive care.
After approximately two years, the following results were observed:
What are the side effects?
The most common side effects of treatment with Cyramza include fatigue, nausea, vomiting, diarrhea, lack of appetite, high blood pressure, swelling in the arms, legs, hands, or feet, pain in the stomach or back, headache, nose bleeds, fever, inflammation of the lining of the mouth, trouble falling or staying asleep, infections, hair loss, bleeding in the stomach or intestine, bleeding in the gums, bleeding in the lungs or respiratory tract, low levels of sodium, calcium potassium, or albumin (protein) in the blood, low platelet count, low white blood cell count, low red blood cell count, buildup of fluid in the belly, too much protein in the urine, increased levels of liver enzymes, and thyroid gland problems.
Treatment with Cyramza can cause side effects that can become serious or life-threatening, and may lead to death. Some of the serious side effects related to Cyramza include blocked digestive tract, pneumonia, collapsed lung, skin infection, severe bleeding (including in the stomach or intestine), having wounds that do not heal, severe high blood pressure, blood clots, and development of holes in the stomach or intestine. Additionally, problems can arise with the heart, kidneys, the thyroid, and the brain. Cyramza may worsen certain types of liver disease. Reactions related to the infusion may also occur. Patients should report any symptoms to their healthcare provider, who can then initiate actions to limit or reverse the side effects.
For a more complete list of possible side effects, see the full prescribing information.
Additional information
Manufacturer
Eli Lilly
Approval
FDA and EMA
Links to drug websites
- US: https://www.cyramza.com/
- Europe: https://www.ema.europa.eu/en/medicines/human/EPAR/cyramza
Last updated on January 16, 2023


